necessary materials and equipment required for galvanic cathodic protection of underground pipes and fittings must be considered. First you must understand what corrosion is and how it occurs.
TYPES OF CORROSION
Man has had corrosion problems to contend with ever since he started making articles out of metal. For thousands of years, the only fact known about corrosion was that it would affect some metals more than others. For example, iron, one of the most abundant and useful metals, corrodes very much; whereas metals such as gold, platinum, and silver corrode very little. Later, men began to study corrosion to find out what caused it. As might be expected, many theories were proposed to explain corrosion and its causes. Among the many theories, the electrochemical theory is most generally accepted as an explanation of corrosion.
The electrochemical theory of corrosion is best explained by the action that takes place in a galvanic cell. A galvanic cell can be produced by placing two dissimilar metals in a suitable electrolyte, as shown in figure 7-12. The resulting electrochemical reaction develops a potential difference between these metals. This causes one metal to be negative or anodic and the other metal to be positive or cathodic. In a dry cell battery, the zinc case is the anode and the carbon rod the cathode. Now, when an external electrical circuit is completed, current flows from the zinc case into the electrolyte, taking with it particles of zinc. This is an example of galvanic corrosion of the zinc case. It is this electrochemical action that illustrates the electrochemical theory.
Corrosion may be divided into several types, such as uniform corrosion, localized corrosion, and compositional corrosion. Each type will be explained in the following paragraphs.
Uniform Corrosion
Uniform corrosion is caused by direct chemical attack. An example of this type of corrosion is zinc exposed to hydrochloric acid. If you examine the surface of zinc in a solution of hydrochloric acid, you will find that the entire surface is corroding. Furthermore, if the zinc is left in the acid long enough, it will be dissolved by the acid.
Localized Corrosion
Localized corrosion is caused by the electrolytic action of a galvanic cell. A local galvanic action is set up when there is a difference of potential between the areas on a metallic surface that is an electrolyte. Localized corrosion may be in the form of pits, pockets, or cavities due to the deterioration or destruction of metal.
Localized corrosion may develop under a number of various conditions when different types
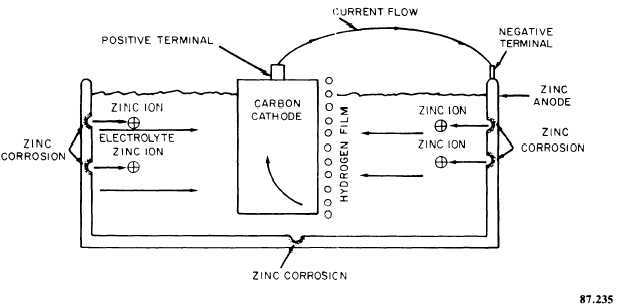
Figure 7-12. - Galvanic cell showing internal galvanic action.
Continue Reading