As the air passes over the oil reservoir, most of the particles will not be able to make the turn, and they will hit the oil and be trapped.
As the air continues upward and passes through the filter element, the smaller particles that bypassed the oil will be trapped.
The air keeps the element soaked with oil by creating a fine spray, as it passes the reservoir.
The air then makes another 180-degree turn and enters the carburetor.
The dry-type air cleaner passes the incoming air through a filtering medium before it enters the engine. The air filter contains a ring of filter material (fine-mesh metal threads or ribbons, pleated paper, cellulose fiber, or polyurethane), as shown in figure 4-16. These types of filter materials provide a fine maze that traps most of the airborne particles.
The air cleaner also muffles the noise of the intake air through the carburetor or fuel injection system, manifold, and valve ports. This noise would be very noticeable if it were not for the air cleaner. In addition the air cleaner acts as a flame arrester in case the engine backfires through the intake manifold. The air cleaner prevents the flame from escaping and igniting gasoline fumes outside the engine.
Q1. What fuel additive is used to prevent engine ping or knock?
Q2. What is the measurement of the ability of gasoline to resist knock or ping?
Q3. What device is used to prevent the accidental use of leaded fuel in a vehicle designedfor unleaded fuel?
Q4. What are the two types of air cleaners currently being used?
PRINCIPLES OF CARBURETION
LEARNING OBJECTIVE: Describe the operating systems and principles of a simple carburetor and a computerized controlled carburetor. Identify the different carburetor accessories and their functions. Identify and describe possible carburetor troubles and quick system checks.
The principles of carburetion are presented so you may better understand the inner workings of a carburetor and how the other components of the fuel system function to provide a combustible mixture or air and fuel to the engine cylinders.
Air is composed of various gases, mostly nitrogen and oxygen (78 percent nitrogen and 21 percent oxygen by volume). These gases are, in turn, made up of tiny particles called molecules. All substances, whether solid, liquid, or gas, are made up of molecules. In solids, such as ice or iron, the particles are held closely together so that they seem to have no motion. In liquids, the molecules are not held together tightly, so they can move freely with respect to each other. In gases, there is still less tendency for the molecules to bond; therefore, the molecules can move quite freely. The molecules of gas are attracted to the earth by gravity or by their weight. It is the combined weight of the countless molecules in the air that make up atmospheric pressure.
Evaporation is the changing of a liquid to a vapor. The molecules of the liquid not being closely tied together are constantly moving among themselves. Any molecule that moves upward with sufficient speed will jump out of the liquid and into the air. This process will cause the liquid to evaporate over a period of time. The rate of evaporation is dependent on the following:
TEMPERATURE. The rate of movement of the molecules increase with temperature. Because of this,
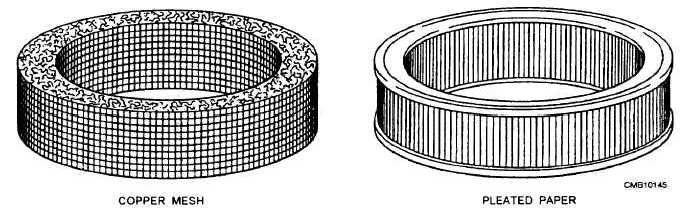
Figure 4-16. - Dry-type air filter.
Continue Reading