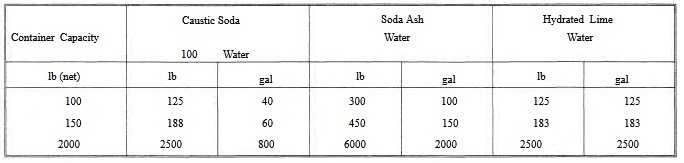
Figure 7-11. - Motor driven hypochlorinator with fully automatic control.
For emergencies, a standby alkali absorption system with a suitable tank should be provided. The alkali should be stored so a solution can be readily prepared. Chlorine should be passed into the solution through a suitable connection properly submerged and weighted to hold it secure. DO NOT IMMERSE CHLORINE CONTAINERS IN SOLUTION. Table 7-1 shows a chemical equivalent. Try to use quantities in excess to allow absorption.
HYPOCHLORITES
To prevent accidents caused by the corrosive action of hypochlorite solutions, use vitreous crocks or steel tanks lined with rubber or chlorine-resistant plastic as solution containers. Store calcium hypochlorite in a dry, cool location, and keep the cans sealed. Wear rubber gloves and protective aprons when preparing and handling hypochlorite solutions.
The liquid CO, recarbonization system uses carbon dioxide gas to lower the pH of softened and settled potable water. This unit is designed for operation between -10F and +3F that corresponds to pressure of 242.8 psig and 306.8 psig. The pressure vessel is designed for temperatures as low as -20F and up to 350 psig. The unit comes with or without a vaporizer, depending on the quantity of CO, required. (If large amounts of CO2 are removed, it is possible to cause the temperature to go below -20F.)Once the unit is installed and in proper operation, it requires little attention. At the beginning and end of each day, the operator should check the pressure and liquid level gauges. The supply valve should be turned off when the unit is not in use. The standard unit is equipped with
Table 7-1. - Recommended Alkaline Solutions for Absorbing Chlorine