It must act as a capacitor (voltage stabilizer) that smoothes current flow through the electrical system.
It must store energy (electricity) for extended periods.
The type of battery used in automotive, construction, and weight-handling equipment is a lead-acid cell-type battery. This type of battery produces direct current (dc) electricity that flows in only one direction. When the battery is discharging (current flowing out of the battery), it changes chemical energy into electrical energy, thereby, releasing stored energy. During charging (current flowing into the battery from the charging system), electrical energy is converted into chemical energy. The battery can then store energy until the vehicle requires it.
Battery Construction
The lead-acid cell-type storage battery is built to withstand severe vibration, cold weather, engine heat, corrosive chemicals, high current discharge, and prolonged periods without use. To test and service batteries properly, you must understand battery construction. The construction of a basic lead-acid cell-type battery is as follows:
Battery element
Battery case, cover, and caps
Battery terminals
Electrolyte
BATTERY ELEMENT. - The battery element is made up of negative plates, positive plates, separators, and straps (fig. 2-3). The element fits into a cell compartment in the battery case. Most automotive batteries have six elements.
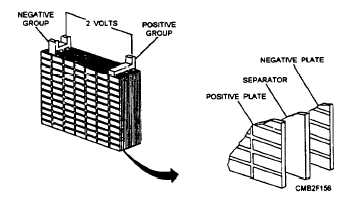
Figure 2-3. - Battery element.
Each cell compartment contains two kinds of chemically active lead plates, known as positive and negative plates. The battery plates are made of GRID (stiff mesh framework) coated with porous lead. These plates are insulated from each other by suitable separators and are submerged in a sulfuric acid solution (electrolyte).
Charged negative plates contain spongy (porous) lead (Pb) which is gray in color. Charged positive plates contain lead peroxide (PbO2 ) which has a chocolate brown color. These substances are known as the active materials of the plates. Calcium or antimony is normally added to the lead to increase battery performance and to decrease gassing (acid fumes formed during chemical reaction). Since the lead on the plates is porous like a sponge, the battery acid easily penetrates into the material. This aids the chemical reaction and the production of electricity.
Lead battery straps or connectors run along the upper portion of the case to connect the plates. The battery terminals (post or side terminals) are constructed as part of one end of each strap.
To prevent the plates from touching each other and causing a short circuit, sheets of insulating material (microporous rubber, fibrous glass, or plastic- impregnated material), called separators, are inserted between the plates. These separators are thin and porous so the electrolyte will flow easily between the plates. The side of the separator that is placed against the positive plate is grooved so the gas that forms during charging will rise to the surface more readily. These grooves also provide room for any material that flakes from the plates to drop to the sediment space below.
BATTERY CASE, COYER, AND CAPS. - The battery case is made of hard rubber or a high - quality plastic. The case must withstand extreme vibration, temperature change, and the corrosive action of the electrolyte. The dividers in the case form individual containers for each element. A container with its element is one cell.
Stiff ridges or ribs are molded in the bottom of the case to form a support for the plates and a sediment recess for the flakes of active material that drop off the plates during the life of the battery. The sediment is thus kept clear of the plates so it will not cause a short circuit across them.
The battery cover is made of the same material as the container and is bonded to and seals the container. The cover provides openings for the two battery posts and a cap for each cell.
Continue Reading