Charles Boyle, an English scientist, was among the first to experiment with the pressure-volume relationship of gas. During an experiment when he compressed a volume of air, he found that the volume decreased as pressure increased, and by doubling the force exerted on the air, he could decrease the volume of the air by half (fig. 3-50).
Temperature is a dominant factor affecting the physical properties of gases. It is of particular concern in calculating changes in the state of gases. Therefore, the experiment must be performed at a constant temperature. The relationship between pressure and volume is known as Boyle's law. Boyle's law states when the temperature of a gas is constant, the volume of an enclosed gas varies inversely with pressure.
Boyle's law assumes conditions of constant temperature. In actual situations this is rarely the case. Temperature changes continually and affects the volume of a given mass of gas.
Charles's Law
Jacques Charles, a French physicist, provided much of the foundation for modem kinetic theory of gases. Through experiments, he found that all gases expand and contract proportionally to the change in absolute temperature, providing the pressure remains constant. The relationship between volume and temperature is known as Charles's law. Charles's law states that the volume of a gas is proportional to its absolute temperature if constant pressure is maintained.
PNEUMATIC GASES
Gases serve the same purpose in pneumatic systems as liquids serve in hydraulic systems. Therefore, many of the same qualities that are considered when selecting a liquid for a hydraulic system must be considered when selecting a gas for a pneumatic system.
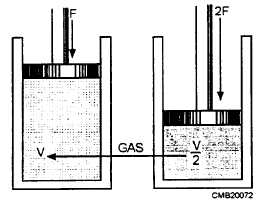
Figure 3-50. - Gas compressed to half its original volume by a doubled force.
Qualities
The ideal fluid medium for a pneumatic system must be a readily available gas that is nonpoisonous, chemically stable, free from any acids that can cause corrosion of system components, and nonflammable. It should be a gas that will not support combustion of other elements.
Gases that have these desired qualities may not have the required lubricating power. Therefore, lubrication of the components must be arranged by other means. For example, some air compressors are provided with a lubricating system, some components are lubricated upon installation or, in some cases, lubrication is introduced into the air supply line (in- line oilers).
Two gases meeting these qualities and most commonly used in pneumatic systems are compressed air and nitrogen. Since nitrogen is used very little except in gas-charged accumulators, we will only discuss compressed air.
Compressed Air
Compressed air is a mixture of all gases contained in the atmosphere. However, in this manual it is referred to as one of the gases used as a fluid medium for pneumatic systems. The unlimited supply of air and the ease of compression make compressed air the most widely used fluid for pneumatic systems. Although moisture and solid particles must be removed from the air, it does not require the extensive distillation or separation process required in the production of other gases.
Compressed air has most of the desired charac- teristics of a gas for pneumatic systems. It is nonpoisonous and nonflammable but does contain oxygen which supports combustion. The most undesirable quality of compressed air as a fluid medium for a pneumatic system is moisture content. The atmosphere contains varying amounts of moisture in vapor form. Changes in the temperature of compressed air will cause condensation of moisture in the system. This condensed moisture can be very harmful to the system and may freeze the line and components during cold weather. Moisture separators and air dryers are installed in the lines to minimize or eliminate moisture in systems where moisture would deteriorate system performance.
An air compressor provides the supply of compressed air at the required volume and pressure. In
Continue Reading