SEMICONDUCTORS
A semiconductor is an electrical device that acts as a conductor under certain conditions and as a nonconductor under other conditions. The most popular of all semiconductors is silicon. In its pure state, silicon is neither a good conductor nor insulator. But by processing silicon in the following ways, its conductive or insulative properties can be adjusted to suit just about any need. When a number of silicon atoms are jammed together in crystalline (glasslike) form, they form a covalent (sharing) bond. Therefore, the electrons in the outer ring of one silicon atom join with the outer ring of other silicon atoms, resulting in a sharing of outer ring electrons between all of the atoms. It can be seen in figure 1-4 that covalent sharing gives each atom eight electrons in its outer orbit, making the orbit complete. This makes the material an insulator because it contains more than four electrons in its outer orbit. When certain materials, such as phosphorus, are added to the silicon crystal in highly controlled amounts, the resultant mixture becomes a conductor (fig. 1-5). This is because phosphorus, which has five electrons in forming a covalent bond with silicon (which has four electrons in its outer shell), will yield one free electron per molecule, thus making the material an electrical conductor. The process of adding impurities to a semiconductor is called doping. Any semiconductor material that is doped to yield free electrons is called N-type material.
When boron, which has three electrons in its outer ring, is used to dope the silicon crystal, the resultant
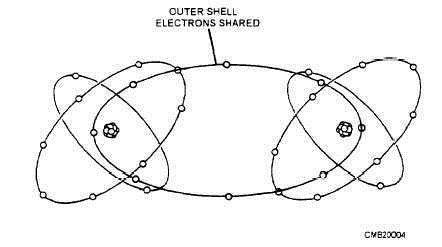
Figure 1-4. - Covalent bonding of silicon.
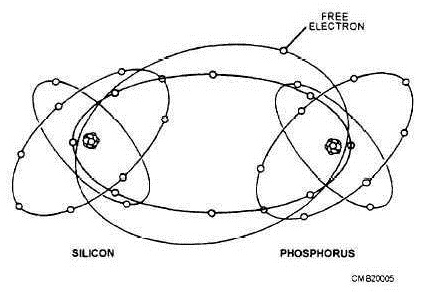
Figure l-5. - Phosphorus-doped silicon.
Continue Reading