For instance, water boils at 212F at atmospheric pressure (14.7 psia), as shown in view A, figure 6-7. The same water boils at 228F if the pressure is raised 5.3 psig (20 psia), as shown in view B, figure 6-7. On the other hand, the same water boils at 32F in a partial vacuum of 29.74 inches of mercury (Hg), as shown in figure 6-8.
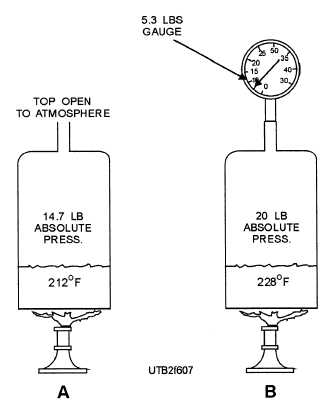
Figure 6-7. - A. Water boils at atmospheric pressure; B. Water boils at 20-psia absolute pressure.
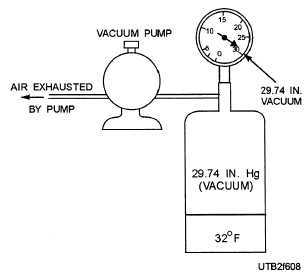
Figure 6-8. - Water boils quicker in a vacuum.
This effect of reduced pressure on the boiling temperature of refrigerants makes the operation of a refrigeration system possible. The pressure temperature relationship chart in figure 6-9 gives the pressures for several different refrigerants.
An increase in the temperature of a refrigerant results in an increase in pressure, and a decrease in temperature causes a decrease in pressure. By the same token, a decrease in pressure results in a corresponding decrease in temperature.
This means that as the pressure of a refrigerant is increased, so is the temperature at which the refrigerant boils. Thus, by regulating the pressure of the refrigerant, the temperature at which evaporation takes place and at which the latent heat of evaporation is used can be controlled.
VAPORIZATION
VAPORIZATION is the process of changing a liquid to vapor, either by evaporation or boiling. When a glass is filled with water, as shown in figure 6-10, and exposed to the rays of the sun for a day or two, you should note that the water level drops gradually. The loss of water is due to evaporation. Evaporation, in this case, takes place only at the surface of the liquid. It is gradual, but the evaporation of the water can be speeded up if additional heat is applied to it. In this case, the boiling of the water takes place throughout the interior of the liquid. Thus the absorption of heat by a liquid causes it to boil and evaporate.
Vaporization can also be increased by reducing the pressure on the liquid, as shown in figure 6-11. Pressure reduction lowers the temperature at which liquid boils and hastens its evaporation. When a liquid evaporates, it absorbs heat from warmer surrounding objects and cools them. Refrigeration by evaporation is based on this method. The liquid is allowed to expand under reduced pressure, vaporizing and extracting heat from the container (freezing compartment), as it changes from a liquid to a gas. After the gas is expanded (and heated), it is compressed, cooled, and condensed into a liquid again.
CONDENSATION is the process of changing a vapor into a liquid. For example, in figure 6-12, a warm atmosphere gives up heat to a cold glass of water, causing moisture to condense out of the air and form on the outside surface of the glass. Thus the removal of heat from a vapor causes the vapor to condense.
Continue Reading