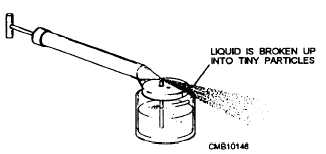
Figure 1-17. - Example of atomization.
the amount of molecules leaving the liquid for a given time will increase, as the temperature increases.
ATMOSPHERIC PRESSURE. As atmospheric pressure increases, the amount of air molecules present over the liquid also increases. The increased presence of air molecules will slow the rate of evaporation. This is because the molecules of liquid will have more air molecules to collide with. In many cases, they will fall back into the liquid after the collision
CLOSED CHAMBER. As evaporation takes place in a closed container, the space above the liquid will reach a point of saturation. When this happens, every molecule of liquid that enters the air will cause another airborne molecule of liquid to fall back.
VOLATILITY. The term volatility refers to how fast a liquid vaporizes. Some liquid vaporizes easily at room temperature. Alcohol, for instance, vaporizes more easily than water. A highly volatile liquid is one that is considered to evaporate easily.
ATOMIZATION (fig. 4-17). Atomization is the process of breaking up a liquid into tiny particles or droplets. When a liquid is atomized, the droplets are all exposed individually to the air. For this reason, atomization greatly increases evaporation by increasing the exposed surface area of the liquid.
The venturi effect (fig. 4-18) is used by the carburetor to mix air with the gasoline. The basic carburetor has an hourglass-shaped tube called a throat. The most constricted part of the throat is called the venturi. A tube, called the discharge nozzle, is positioned in the venturi. The discharge nozzle is connected to a reservoir of gasoline called the float bowl. The negative pressure that exists in the combustion chamber is due to the downward intake stroke of the piston, causing atmospheric pressure to create an air flow through the throat. This air flow must increase temporarily in speed, as it passes through the venturi due to its deceased size. The increased speed of air flow results in a corresponding decrease in pressure within the venturi and at the end of the discharge nozzle. This action permits the atmospheric pressure on the surface of the gasoline in the float bowl to force the gasoline out through the discharge nozzle. This gasoline then sprays and atomizes in the passing air flow to form the air-fuel mixture.
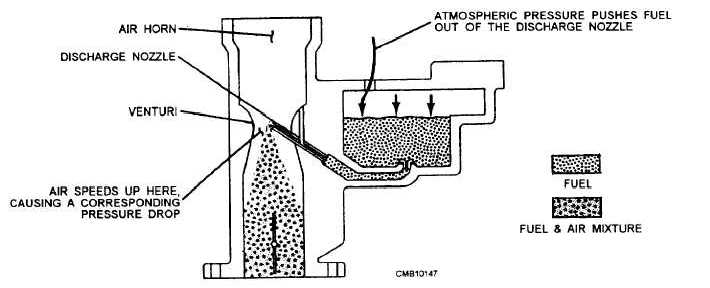
Figure 4-18. - Venturi effect.
Continue Reading