CHAPTER 1 PROPERTIES AND USES OF METAL
In the seabees, Steelworkers are the resident experts on the properties and uses of metal. We lay airfields, erect towers and storage tanks, assemble pontoon causeways, and construct buildings. We use our expertise to repair metal items, resurface worn machinery parts, and fabricate all types of metal objects. To accomplish these tasks proficiently, one must possess a sound working knowledge of various metals and their properties. As we learn their different properties and characteristics, we can then select the right type of metal and use the proper method to complete the job. Steelworkers primarily work with iron and steel; however, we also must become familiar with the nonferrous metals coming into use more and more each day. As Steelworkers, we must be able to identify various metals and to associate their individual properties with their proper application or use.
The primary objective of this chapter is to present a detailed explanation of some of the properties of different metals and to provide instruction on using simple tests in establishing their identity.
METAL PROPERTIES
There is no simple definition of metal; however, any chemical element having "metallic properties" is classed as a metal. "Metallic properties" are defined as luster, good thermal and electrical conductivity, and the capability of being permanently shaped or deformed at room temperature. Chemical elements lacking these properties are classed as nonmetals. A few elements, known as metalloids, sometimes behave like a metal and at other times like a nonmetal. Some examples of metalloids are as follows: carbon, phosphorus, silicon, and sulfur.
Although Steelworkers seldom work with pure metals, we must be knowledgeable of their properties because the alloys we work with are combinations of pure metals. Some of the pure metals discussed in this chapter are the base metals in these alloys. This is true of iron, aluminum, and magnesium. Other metals discussed are the alloying elements present in small quantities but important in their effect. Among these are chromium, molybdenum, titanium, and manganese.
An "alloy" is defined as a substance having metallic properties that is composed of two or more elements. The elements used as alloying substances are usually metals or metalloids. The properties of an alloy differ from the properties of the pure metals or metalloids that make up the alloy and this difference is what creates the usefulness of alloys. By combining metals and metalloids, manufacturers can develop alloys that have the particular properties required for a given use.
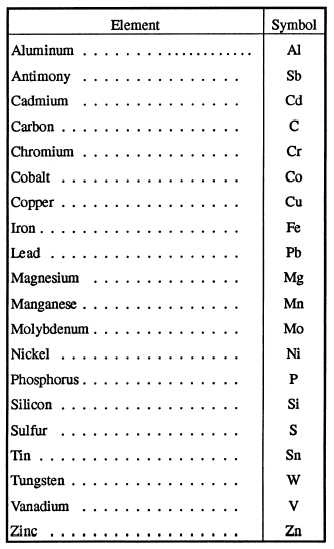
Table 1-1 is a list of various elements and their symbols that compose metallic materials.
Table 1-1. - Symbols of Base Metals and Alloying Elements