settle are called colloidal particles. Colloidal particles are often removed in the biological treatment units. They may also be removed by chemical treatment followed by sedimentation.
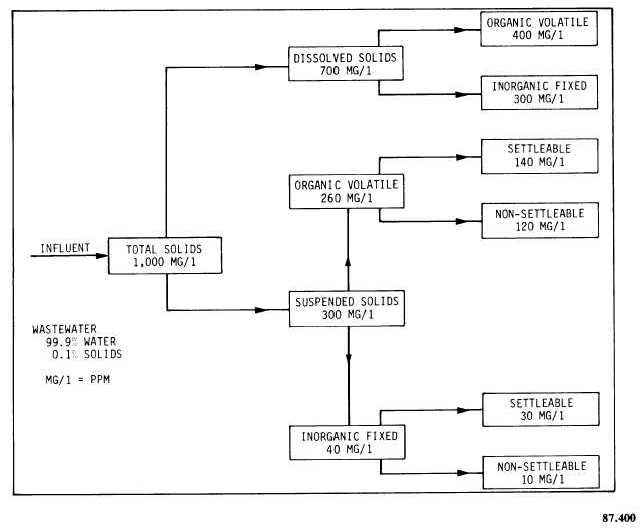
All the solids discussed above may be either organic or inorganic. Organic solids always contain carbon and hydrogen and when ignited to high temperatures (500C to 600C) burn to form carbon dioxide, water, and sometimes various other compounds. The burning or volatilization of organic solids has led to the term volatile solids. All solids that burn or evaporate at 500C to 600C are called volatile solids. These solids serve as a food source for bacteria and other living forms in a wastewater treatment plant. Most organic solids in municipal waste originate from living plants or animals.
Those solids that do not burn or evaporate at 500C to 600C, but remain as a residue, are called fixed solids. Fixed solids are usually inorganic in nature and may be composed of grit, clay, salts, and metals. Most inorganic solids are from nonliving sources. Table 10-4 summarizes the types and amounts of the solids discussed in the preceding paragraphs.
CHEMICAL CHARACTERISTICS
The chemical characteristics of wastewater of special concern to the Utilitiesman are pH, DO (dissolved oxygen), oxygen demand, nutrients, and toxic substances.
pH
The term pH is used to describe the acid or base properties of water solutions. A scale from
Table 10-4.-Solids of a Typical Domestic Wastewater