7. Record the total ml of acid required to reach this end point. This includes the ml consumed in the phenolphthalein alkalinity titration and that consumed in the methyl orange titration.
CALCULATIONS - The phenolphthalein (PT) alkalinity is calculated as ppm of calcium carbonate by multiplying the ml of acid used in the phenolphthalein titration by 20.
ppm PT alkalinity as calcium carbonate = ml of acid used in step x 20
The total alkalinity, as ppm of calcium carbonate, is found by multiplying the total number of ml of acid used (Step 6 above) by 20. This applies to both the methyl orange and the methyl purple procedures.
ppm total alkalinity as calcium carbonate = total ml acid used x 20
The Hardness Test
The titration method for determining water hardness is vastly superior to the old soap test that is slow, tedious, and often may give misleading results. The procedure is based on the fact that when a sample of water is titrated with a solution of EDTA (sodium ethylene, diamine tetra- acetate), calcium and magnesium react with the EDTA to form soluble compounds in which calcium and magnesium are tied up so firmly that they cannot react with other materials. Standard EDTA solution is added to a water sample and the end point is detected by an indicator that is red in the presence of calcium and magnesium ions and blue in their absence. A total hardness test set (EDTA) is shown in figure 7-16.
REAGENTS - The following reagents are used in testing for water hardness:
EDTA solution
Hardness indicator powder
Hardness buffer
Hardness reagent
PROCEDURES - In determining water hardness, here is the procedure to follow.
1. Place a 50-ml sample in a 250-ml Erlenmeyer flask.
2. Add 1 dipper of hardness indicator powder.
3. Add 0.50 ml of hardness buffer to hold the pH at around 10. The color of the mixture will be red if any hardness is present.
4. Add the hardness reagent from a burette until the red color just disappears, giving way to a pure blue.
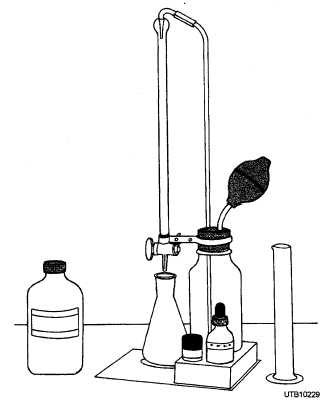
Figure 7-16. - Total hardness test set (EDTA).
CALCULATIONS - The burette reading in ml is multiplied by 20 to give the total hardness.
ppm total hardness as calcium carbonate = ml burette readingg x 20
The Chloride Test
The purpose of the chloride test is to measure the amount of chloride ions and common salt (NaCl) in water. This test also indicates the presence of possible sewage pollution.
REAGENTS - The reagents used in making the chloride tests are as follows:
Phenolphthalein Indicator
Methyl Orange Indicator
Potassium Chromate
Silver Nitrate Standard
Aluminum Hydroxide
Sulfuric Acid (1 to 3)
PROCEDURES - When making a chloride test, follow the procedure below.
Continue Reading