Battery caps either screw or snap into the openings in the battery cover. The battery caps (vent plugs) allow gas to escape and prevent the electrolyte from splashing outside the battery. They also serve as spark arresters (keep sparks or flames from igniting the gases inside the battery). The battery is filled through the vent plug openings. Maintenance-free batteries have a large cover that is not removed during normal service.
CAUTION
Hydrogen gas can collect at the top of a battery. If this gas is exposed to a flame or spark, it can explode.
BATTERY TERMINALS. - Battery terminals provide a means of connecting the battery plates to the electrical system of the vehicle. Either two round post or two side terminals can be used.
Battery terminals are round metal posts extending through the top of the battery cover. They serve as connections for battery cable ends. Positive post will be larger than the negative post. It may be marked with red paint and a positive (+) symbol. Negative post is smaller, may be marked with black or green paint, and has a negative (-) symbol on or near it.
Side terminals are electrical connections located on the side of the battery. They have internal threads that accept a special bolt on the battery cable end. Side terminal polarity is identified by positive and negative symbols marked on the case.
ELECTROLYTE. - The electrolyte solution in a fully charged battery is a solution of concentrated sulfuric acid in water. This solution is about 60 percent water and about 40 percent sulfuric acid.
The electrolyte in the lead-acid storage battery has a specific gravity of 1.28, which means that it is 1.28 times as heavy as water. The amount of sulfuric acid in the electrolyte changes with the amount of electrical charge; also the specific gravity of the electrolyte changes with the amount of electrical charge. A fully charged battery will have a specific gravity of 1.28 at 80F. The figure will go higher with a temperature decrease and lower with a temperature increase.
As a storage battery discharges, the sulfuric acid is depleted and the electrolyte is gradually converted into water. This action provides a guide in determining the state of discharge of the lead-acid cell. The electrolyte that is placed in a lead-acid battery has a specific gravity of 1.280.
The specific gravity of an electrolyte is actually the measure of its density. The electrolyte becomes less dense as its temperature rises, and a low temperature means a high specific gravity. The hydrometer that you use is marked to read specific gravity at 80F only. Under normal conditions, the temperature of your electrolyte will not vary much from this mark. However, large changes in temperature require a correction in your reading.
For EVERY 10-degree change in temperature ABOVE 80F, you must ADD 0.004 to your specific gravity reading. For EVERY 10-degree change in temperature BELOW 80F, you must SUBTRACT 0.004 from your specific gravity reading. Suppose you have just taken the gravity reading of a cell. The hydrometer reads 1.280. A thermometer in the cell indicates an electrolyte temperature of 60F. That is a normal difference of 20 degrees from the normal of 80F. To get the true gravity reading, you must subtract 0.008 from 1.280. Thus the specific gravity of the cell is actually 1.272. A hydrometer conversion chart similar to the one shown in figure 2-4 is usually found on the hydrometer. From it, you can obtain the specific gravity correction for temperature changes above or below 80F.
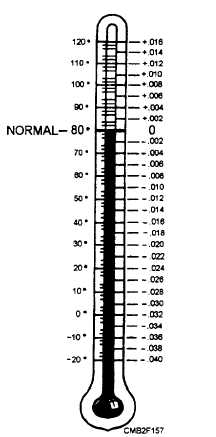
Figure 2-4. - Hydrometer conversion chart.
Continue Reading